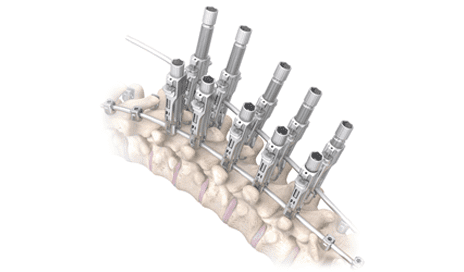
OrthoPediatrics, headquartered in Warsaw, Indiana, recently received 510(k) FDA clearance for its RESPONSE 5.5/6.0 Spine System, which is designed to help surgeons surgically correct scoliosis in pediatric patients.
A news release from the company notes that the system features low-profile implants that accept 5.5-mm or 6.0-mm-diameter rods and intuitive instrumentation to promote ease of use in pediatric anatomy. The system also features the Jiminy™ Rod Reducer instrument, which is designed to snap on and off easily and enables each surgeon to perform reduction and de-rotation techniques for the most complex spinal deformities in pediatric patients.
For more information, visit OrthoPediatrics.
[Source(s): OrthoPediatrics, PRWeb]