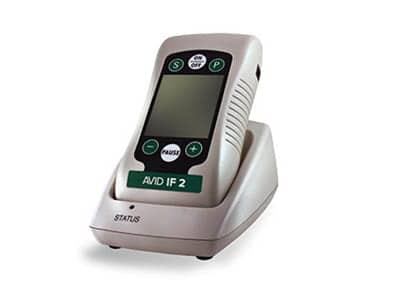
The Avid IF 2 is a noninvasive device built to help manage pain symptoms, and recently introduced by Irvine, Calif-based VQ OrthoCare. The Avid IF 2 is designed as a portable interferential device that provides safe and effective pain management for acute pain and chronic pain, and to help relax muscle spasms, maintain range of motion, and increase circulation.
VQ OrthoCare describes the Avid IF 2 as an electrical stimulator that generates small pulses of current that are delivered through lead wires to stimulating electrodes placed on the patient’s skin. The pulses reportedly pass through the skin and activate underlying nerves. The company further states that the Avid IF 2 is designed to activate sensory nerves at lower levels of stimulation, producing a tingling sensation that blocks out pain messages. Motor nerves are activated a higher levels of stimulation, resulting in increased circulation, reduced muscle spasms, and promotes recovery.
A media release from VQ OrthoCare states that interferential therapy is a recommended treatment option by Official Disability Guidelines (ODG), which positions the Avid IF 2 as an effective pain management protocol. The device is said to allow for web-based compliance reporting through the recording and uploading of detailed usage data to a secure website via a cradle with modem. This data assesses the patient’s progress and assists the treating physician in determining further treatment.
The Avid IF 2 is available by prescription only and is offered as a system complete with non-sterile, reusable electrodes, lead wire, and batteries.
[Source: VQ OrthoCare)