
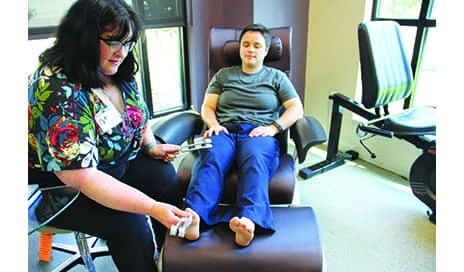
Physical therapist performs vibration therapy using tuning forks. Benefits include: relaxation of body tissues, relieves spasms and pain, improves blood circulation and decreases symptoms associated with chemo-induced peripheral neuropathy. Another use of tuning forks is reflexology using acupressure points.
By Pam Veale, PT
Peripheral neuropathy has been defined as a dysfunction of the sensory, motor, or autonomic nervous system. While other forms of neuropathy may exist, such as diabetic neuropathy or radiculopathy, the most commonly experienced neuropathy in oncology patients is chemotherapy-induced peripheral neuropathy (CIPN) from chemotherapy neurotoxicity. CIPN can be a motor or sensory neuropathy, usually affecting the hands and feet. It is most commonly manifested as a “distal sensory neuropathy, which is characterized by pain, numbness, tingling, and reduced functional capacity in the extremities. Other symptoms include paresthesia, ataxia, impaired vibration and joint position sense, and loss of tendon reflexes.1 As per Hershman et al, in most cases, the chemotherapy causes damage to the nerve axons, cellular metabolism, and nerve growth factor.1 These all seem to be a part of causing the potentially irreversible damage of CIPN.1 The exact mechanism of damage, however, varies with different types of chemotherapy agents.
While most cancer treatment focuses on increasing the length or quantity of life, physical and occupational therapists—along with other providers such as acupuncturists, naturopathic providers, and massage therapists—focus therapeutic attention to preserving the quality of a patient’s life. Advances in cancer treatments have resulted in significantly improved survival for many cancer diagnoses, but these treatments have also resulted in ongoing functional impairments for many survivors.2 Caveletti reported that while there is improvement in cancer survival statistics, there are also long-term side effects that limit the functional performance and overall quality of life of the survivors, both during treatment and persisting for years afterward.2
Common medications that can cause CIPN include taxanes, platinum-based drugs, and vinca alkaloids.3 This neurotoxicity is not only potentially painful and distressing to the patient, but has been shown to negatively affect the patient’s functional abilities, safety, and ability to participate in oncology treatment. Increased CIPN toxicities past Grade II in the CTCAE V.4 toxicity criteria are often grounds for dose-reducing or possibly discontinuing a chemotherapy that may have been working effectively to fight the cancer.1 Hershman reported that even mild dose reductions can limit the chemo’s effectiveness,1 as CIPN can be “a dose-limiting side effect that leads to suboptimal cancer treatment and may also adversely affect both the survival outcome and long-term quality of life.”1
Studies showed that 80% of the subjects had at least grade I neuropathy, 34% had Grade 2 to 3 neuropathy, and 26% had dose delays or reductions of the chemo.1 For this reason, therapeutic efforts to prevent or manage CIPN would not only enhance quality of life and safety, but would preserve a patient and doctor’s treatment choices and chance for cancer survival.1 CIPN can be chronically limiting, as after following subjects for 1 year, 67% had continued numbness in hands and feet, and 27% still reported severe symptoms. Symptoms persisted for most patients even 2 years after finishing the chemo.1 In addition to these physical costs, research studies showed the large financial impact of CIPN, resulting in not only higher healthcare costs, up to triple those of controls, but also indirect costs of $4,000/episode in lost work and caregiver costs.2

PT applies custom modulating electrical waveform unit that stimulates and heals damaged nerves. The goal is to provide relief of nociceptive symptoms associated with chemo-induced peripheral neuropathy, such as tingling, numbness and pain.
Functional Impairments Due to CIPN
Falls and mobility deficits
According to Tofthagan, Overcash, and Kip,4 patients on chemotherapy are more than two times more likely to fall than those not receiving chemo.4 They also reported the CIPN symptoms of diminished sensation, impaired proprioception, and strength deficits in the legs can each cause difficulties with walking and interfere with balance, therefore increasing the risk for falls.4 The compounding factors of fatigue, decline in performance status, and anemia also increase the likelihood for falls.4 Tofthagan et al’s study concluded that “risk for falls increases with each cycle of chemo,”4 and those patients with more intense levels of CIPN symptoms showed higher risk for falls4; as well as “Loss of sensation, loss of proprioception and weakness in the muscles of the lower extremities are common signs and symptoms of peripheral neuropathy, regardless of etiology. Diminished sensation and loss of neuromuscular control…eventually interfere with balance and gait.”4
One review showed cancer patients at least two times more likely to fall and reported 50% of advanced cancer patients fell in the 6 months following treatment.5 Gewandter et al’s study was the first to follow falls specifically due to CIPN. They found that even including younger survivors, those with CIPN have up to a 40% greater fall risk than healthy adults over 65 years of age.5 Although the symptoms vary between types of chemo, all can contribute to the increased falls noted in patients with CIPN. Therapists commonly use balance and mobility tests such as the Berg balance, Timed Up and Go, and Single Leg stance tests to verify levels of balance dysfunction caused by CIPN.
Other functional impairments
Falls are not the only functional impairment noted with CIPN. A larger study included 421 patients, with 11.9% having falls but 26.6% also reporting other functional impairments.4 Other functional impairments noted were limitations in ability to shop, bathe, manage money, walk across the room, and do light housework.5 Occupational therapists often assess fine motor coordination, strength, and ADL function with various screening tools such as the Barthel Index, grip strength testing, peg board testing, as well as many other validated tools to assess patients with CIPN impairments.
Sensory deficits and pain
The common symptom of sensory deficits, as seen with platinum-based chemotherapies, most frequently affected ADLs and other functional disabilities, however with slightly fewer falls noted.5 Semmes Weinstein monofilaments, tuning forks, and other sensation testing tools are used to assess levels of sensation deficits and monitor changes in these symptoms. The CIPN pain symptoms are also evaluated by visual analog scales, and numerous pain inventories and questionnaires. The lack of reliable and consistently accurate assessment tools is another avenue needing to be validated to help determine severity of CIPN and efficacy of the treatments.
Management Options for CIPN
Many types of pharmaceutical and non-pharmaceutical options are being utilized in the treatment of CIPN. Non-pharmacological treatments utilized by therapists may be low in risk and cost, but further research is needed to better validate the results. Many previous studies have been done on diabetic neuropathy, which has a different pathogenesis than CIPN with no proven correlation of the results to the CIPN population. One research study by Pachman et al6 was a helpful resource identifying many available options with positive initial results both for non-CIPN conditions and CIPN. The purpose of the study was to bring the best available evidence on existing treatment options to healthcare providers for clinical decision-making, and to demonstrate the need for further research to validate their effectiveness. They outlined the use of medications, topical treatments, and supplements in addition to the non-pharmaceutical options of acupuncture, electrical stimulation, and exercise.6
Examples of medications used:
Antidepressants, anticonvulsants, muscle relaxants, opioids, lidocaine patches.3 Other therapies include topical treatments such as capsaicin patches, and naturopathic supplements such as vitamin B6, vitamin E, and acetyl-L-carnitine.3
Exercise
Exercise is one of the hallmark treatments of physical and occupational therapists for the functional impairments resulting from CIPN. The weakness, fatigue, and neuromuscular deficits seen in CIPN are often treated with exercises for strengthening, proprioceptive stimulation, fine motor and gross motor coordination, and balance training exercises. While there is no evidence that exercise improves CIPN symptoms, it has been shown to reduce falls and improve performance status and overall quality of life.6 Patient education on skin care, foot/hand safety with impaired sensation, use of assistive devices to aid in proprioception, ADLs, and gait safety are also important aspects of therapy treatment for CIPN.
Electrotherapy and other modalities
Many patients utilize traditional TENS units for pain relief, but there are also other specialized electrical modalities designed for use with peripheral neuropathy that provide custom-tailored electrical impulse for nerve stimulation. They use different custom waveforms and frequencies to target the symptoms and neurotoxic damage involved in peripheral neuropathy. More research is needed to validate these modalities, but there are several recent (2016) studies showing promising results from one of the therapy modalities.7 Therapists have historically used infrared light therapies, which also have shown some benefit in research studies. These electrical stimulation and other modalities may not cure CIPN, but can often allow a patient with CIPN to tolerate ambulation and improved ADL function, therefore preserving functional performance status to allow continued oncology treatment. While there was inconclusive evidence in some of the studies for the electrotherapy and exercise treatments on reducing CIPN deficits, there were improvements in subjective pain and physical function following these treatments.6
Acupuncture
There have been several studies that showed acupuncture had some significant subjective and functional improvement with no adverse effects. Results of one study showed 67% of patients reporting CIPN improvement with acupuncture, as compared to 40% in the medication group. In a different study of five patients, all “reported an improvement in sensation, gait, and balance, and were able to decrease their doses of pain medication.”6 This last study reported by Pachman et al was of great interest in that although a small sample size was used, it correlated the acupuncture treatment to improvements in functional impairments associated with CIPN. Another study showed “82% of patients reported that the acupuncture treatment improved CIPN symptoms.”7 In the study, 35% to 42% also reported one or more additional benefits such as better sleep, less medication needed, less stress, and improved mood.8
Acupuncture as a non-pharmaceutical treatment option was also appreciated by patients desiring relief from symptoms without the need to take more medication. Longevity of acupuncture results in CIPN was demonstrated for 6 months in four out of five patients in one case series reviewed.8 Another form of acupuncture called sonopuncture, using vibration instead of needles, has been used for multiple conditions with subjective patient-reported improvement, but there is a lack of research studies confirming sonopuncture’s effectiveness. Massage and energy work therapies have some research validating benefits for pain and many anecdotal reports of decreasing CIPN symptoms, but as with most non-pharmacological treatments for CIPN, further research is needed.
Whole body vibration
Another treatment option with some preliminary research is whole body vibration (WBV). Most of the research however, was with diabetic neuropathy patients using a vibration platform. Kessler and Hong performed a pilot study showing significant reduction in both acute and chronic neuropathy pain using WBV.9 As mentioned previously, there is no specific research showing the reliability of similar results in the CIPN population, but this possible treatment has had anecdotal reports of improvement in patients with CIPN. A potential contraindication of WBV use in the cancer patient population would be if there were a presence of bone metastases. As many cancer patients receiving neurotoxic chemotherapies have cancer types that often metastasize to bone, this could limit WBV use for many CIPN patients.
The use of tuning forks as another vibration tool is already validated for vibratory sensation assessment. This localized vibration administration using tuning forks would eliminate the contraindication in those with bony metastasis. Tuning fork vibration on acupuncture points for CIPN is a combination of acupressure and vibratory stimulation that has anecdotally positive results, but needs further research for validation as an evidenced-based practice. This treatment is low-cost, time-efficient, and can be easily taught to patients for ongoing self care. In the current medical reimbursement climate limiting the number of therapy visits, identifying treatments such as self-vibratory acupressure, that patients could continue at home, would be a financially feasible way to reduce ongoing functional impairments of CIPN.
Laser treatment with low level energy laser (LLEL)
This device is another treatment modality used by therapists for CIPN. The mechanism by which LLEL affects cells is not well-understood, but it seems to be based on bio-stimulation.10 It is believed that low-level radiation is absorbed by intracellular photoreceptors in the membrane of the mitochondria. Low level energy laser (LLEL) has been shown to repair, regenerate, and accelerate tissue healing. It accelerates functions of cytochrome enzymes, increases ATP and Nitric Oxide (NO), enhances cellular metabolism, and improves microcirculation and resultant nerve function. Recent evidence suggests immune cells play a role in neuropathic pain at the periphery.11
The presence of CIPN as a distressing and potentially life-altering side effect of neurotoxic chemotherapies is well-documented. There has been much research detailing the large impact that CIPN has on patient quality of life, but also the socioeconomic burden and negative impact on ability to receive possibly life-saving chemotherapy treatments when CIPN toxicities become too great to continue treatment.
As physical and occupational therapists, we can utilize many therapeutic techniques and modalities, as well as exercises and patient education, to mitigate these problems of CIPN. By being part of an integrated medical approach, we can assist the medical oncologists to manage toxicities. By keeping the physical performance status level of a patient from declining due to CIPN, this will enable them to continue their oncology treatment plan without dose reductions or delays, which can significantly enhance a patient’s overall chance for survival. Therapists contribute in educating and empowering patients to take an active role in improving their safety and minimizing the effects of CIPN on their daily function. This plays a great role in helping minimize risks of falls, improve strength and function, and reduce further functional decline during oncology treatment. Lastly, by a continued focus on research, we can improve the evidence-based validation of current treatments and develop more effective options for peripheral neuropathy therapeutic interventions. RM
Pam Veale, PT, has been a licensed physical therapist for 27 years. The past 10 years she has focused on oncology rehabilitation at Cancer Treatment Centers of America at Southwestern Regional Medical Center in Tulsa, Okla. Veale received her bachelor’s degree in Physical Therapy from the University of Oklahoma. In addition, she recently completed a Master of Science degree in Health Care Administration from Oklahoma State University. For more information, contact [email protected].
References
1. Hershman DL, Weimer LH, Wang A, et al. Association between patient reported outcomes and quantitative sensory tests for measuring long-term neurotoxicity in breast cancer survivors treated with adjuvant paclitaxel chemotherapy. Breast Cancer Res Treat. 2010;125(3):767–774. doi:10.1007/s10549-010-1278-0.
2. Cavaletti G. Chemotherapy-induced peripheral neurotoxicity (CIPN): What we need and what we know. J Peripher Nerv Syst. 2014;19(2):66–76. doi:10.1111/jns5.12073.
3. Lawenda BD. Complementary Therapies for Chemo-Neuropathy: An Integrative Oncologist’s Bag of Tricks. Manage Side Effects Without Drugs. https://integrativeoncology-essentials.com/2013/06/Complementary.
4. Tofthagen C, Overcash J, Kip K. Falls in persons with chemotherapy-induced peripheral neuropathy. Support Care Cancer. 2011;20(3):583–589. doi:10.1007/s00520-011-1127-7.
5. Gewandter JS, Fan L, Magnuson A, et al. Falls and functional impairments in cancer survivors with chemotherapy-induced peripheral neuropathy (CIPN): A university of Rochester CCOP study. Support Care Cancer. 2013;21(7):2059–2066. doi:10.1007/s00520-013-1766-y.
6. Pachman DR, Watson JC, Lustberg MB, et al. Management options for established chemotherapy-induced peripheral neuropathy. Support Care Cancer. 2014;22(8):2281–2295. doi:10.1007/s00520-014-2289-x.
7. Loprinzi C. Mayo clinic researchers test scrambler therapy for pain. Healio-Hematology/Oncology. http://www.healio.com/hematology-oncology/breast-cancer/news/online/%7B39fb0977-4c06-4d72-8c04-841b6e172bba%7D/mayo-clinic-researchers-test-scrambler-therapy-for-pain. Published January 6, 2016.
8. Visovsky C. Acupuncture for the management of chemotherapy-induced peripheral neuropathy. J Adv Pract Oncol. 2012;3(3). doi:10.6004/jadpro.2012.3.3.6.
9. Kessler NJ, Hong J. Whole body vibration therapy for painful diabetic peripheral neuropathy: A pilot study. J Bodyw Mov Ther. 2013;17(4):518–522. doi:10.1016/j.jbmt.2013.03.001.
10. Parker S. Lasers: In Dentistry. United Kingdom: British Dental Journal; May 1, 2007.
11. Moritz A. Oral laser application. Quintessence Pub Co; 2006:521–539.





CTCA uses the ReBuilder as the first line of treatment for chemo induced peripheral neuropathy since 2007. Check it out at: http://www.rebuildermedical.com.
I am interested in low level laser therapy- after my chemo treatments – I have developed neuropathy – HELP