Standing in abduction maintains hip stability for one child with cerebral palsy.
By Maryann M. Girardi, PT, DPT, ATP
“Your daughter needs hip surgery.” No mother wants to hear that, but that is what the orthopedist told Caroline. Her beautiful daughter Briella would need surgery for bilateral hip dysplasia (displacement) if her hips continued to migrate (become more uncovered by the acetabulum). Caroline was not ready to accept surgery as the only possible treatment and went about finding other options for Briella. Thanks to her perseverance, ultimately a standing program prevents surgery.
Hip Dysplasia and Cerebral Palsy
Hip dysplasia/displacement (also known as neuromuscular hip displacement and spastic hip subluxation) is a condition in which the acetabulum does not fully cover the head of the femur. It is one of the most common orthopedic issues for children with cerebral palsy. It is different from developmental dysplasia of the hip (DDH or congenital hip dysplasia), as most children with cerebral palsy are born with normal hips and develop displacement over time due to muscle imbalance and inadequate weightbearing.1 Children are often asymptomatic, and this condition does not always get diagnosed until the hip has become dislocated and/or painful. This has implications for positioning in all orientations, hygiene, and quality of life, especially if the hip is painful and there is a loss of range of motion in the hip joint.
For children with cerebral palsy, the overall incidence of hip displacement is reported to be 35% and the incidence directly correlates to a child’s Gross Motor Function Classification Scale (GMFCS) level. The incidence of hip displacement increases with the child’s GMFCS level; 0% at Level I to 90% at Level IV.2 The GMFCS is used to describe children’s motor function. The scale comprises five levels starting at Level I (the child walks without restriction in all settings but has limitations in more advanced gross motor skills) up to Level V (the child is dependent for all mobility, and has very limited ability to control posture and extremity movement even with assistive technology).3
Hip Surveillance
Hip surveillance is “the process of monitoring and identifying the critical early indicators of hip displacement.” The program includes both clinical and radiological exams. The standard clinical examination includes identifying GMFCS level and Winters, Gage, Hicks gait type, and hip passive range of motion with attention to the presence of pain. The radiological exam includes supine anterior/posterior (AP) x-ray of the pelvis in a standardized position to enable measurement of the migration percentage (MP).4 It is widely accepted that MP (the percentage of the femoral head that is not covered by the acetabulum as measured on a supine anterior/posterior pelvic x-ray) is the gold standard for evaluating hip status.5 MP values greater than 33% are considered hip subluxation and greater than 40% are considered hip dislocation. An MP greater than 30% indicates the hip is at risk of dislocation and the child should be referred to a pediatric orthopedic surgeon.6
Hip surveillance programs have been effective in early diagnosis of hip dysplasia (displacement), enabling early intervention to prevent painful hip dislocation. The intervention includes therapies, postural management, and surgical procedures. These regional and national programs exist in many countries including many in Europe as well as Australia and parts of Canada. The Cerebral Palsy Follow Up program in Sweden has decreased the incidence of hip dislocation from 10% to 0.4% since starting in 1994.7
While there is no national hip surveillance program in the United States, there are programs through specific care sites including most children’s hospitals and through the California Children’s Services’ Medical Therapy Program. The specifics of each program vary among the care sites. To standardize the care in the US, the American Academy for Cerebral Palsy and Developmental Medicine (AACPDM) and the Pediatric Orthopedic Surgeons of North America (POSNA) have both published guidelines for hip screenings by age and GMFCS levels. These guidelines include a schedule for clinical and radiological examinations based on age, GMFCS level, and MP. Children with CP (or suspected CP) should have their first screening at age 2 and then follow the recommended schedule until skeletal maturity.4,8
Briella’s Story
Briella was born at 34 weeks by emergency C-section after marginal cord/placenta and intra uterine growth restriction was diagnosed at her mother’s 33-week prenatal check-up. Her NICU stay was complicated by a staph infection which cleared, and she was discharged home on day 25. Briella’s parents and healthcare providers suspected cerebral palsy at 11 months, due to her prematurity and severely delayed motor skills. She experienced delayed developmental milestones including rolling inconsistently at 6 months, army crawling at 12 months, unsupported sitting at 14 months, and standing with support at 2-3 years of age.
The suspected diagnosis of CP was confirmed by MRI at 21 months, which revealed PVL, or periventricular leukomalacia (damage to the white matter of the brain), and she was diagnosed with spastic diplegia. GMFCS levels are not able to be determined until 2 years of age, but when it was appropriate for her to be assessed, Briella was determined to be at GMFCS Level III.
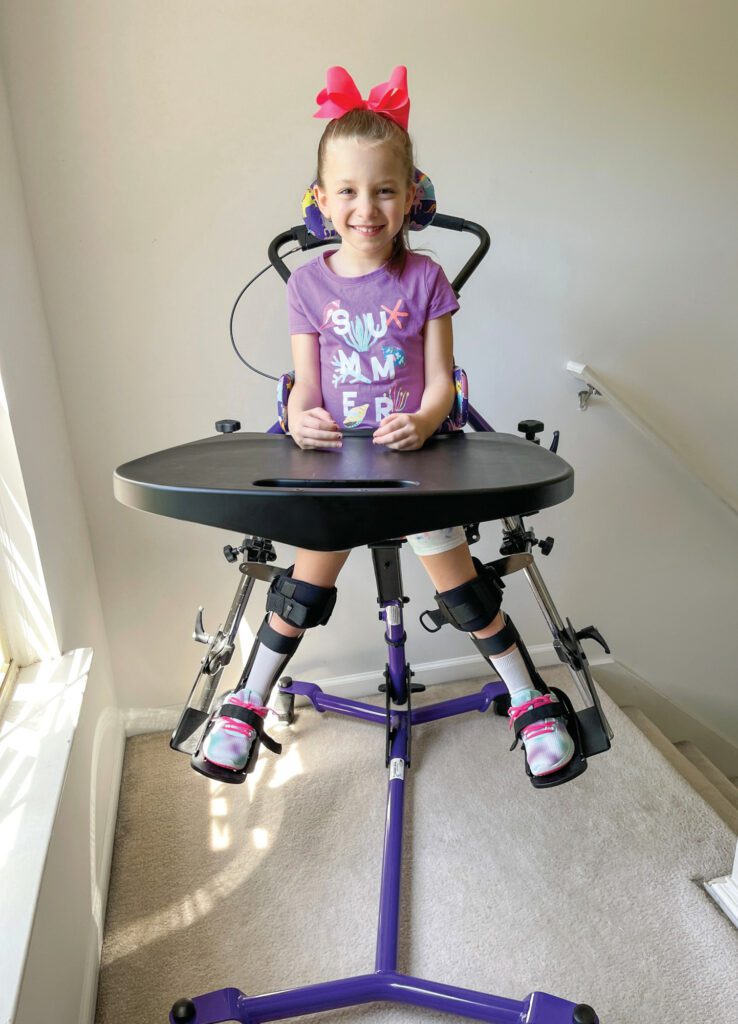
Physical Therapy
Briella started to receive physical therapy services through her local school system starting at 1.5 years old and continues to receive these services today. She also participates in intensive physical therapy programs 1-2 times per year as well as a daily home program with her family.
A major focus of her physical therapy at the beginning was to get her walking. In standing, Briella’s foot ankle posture was compromised, resulting in poor alignment and stability. She required external support to attain and maintain good alignment for walking, so she was referred to an orthopedist for orthotic evaluation. He had her begin wearing ankle foot orthoses (AFOs), and she started to use a posterior walker in physical therapy sessions when she was 2 years old. She received her own walker at 3 years of age but did not like it and used it infrequently at home.
In 2018 at 3.5 years old, Briella underwent a selective dorsal rhizotomy (surgical procedure where selective spinal nerve roots that cause spasticity are cut) for lower extremity spasticity management. A significant decrease in her lower extremity spasticity was noted after the surgery and she continued to participate in physical therapy and started using her walker more at home. By age 5.5 she was using her walker for all community mobility and taking up to 100 steps without it in her home.
Briella was followed by a physiatrist (Doctor of Physical Medicine and Rehabilitation) and her first hip x-rays were taken at 3 years old. These showed the acetabula appeared relatively defined and the femoral heads were normally located with abduction. She did not receive another x-ray until January 2020 at 5.5 years old when she was referred to an orthopedist. That x-ray showed MPs of left 25% and right 30%, which were comparable to the 2018 study.
A Candidate for Surgery
Briella’s next orthopedic visit was in October 2020 at 6 years old, at which time the x-ray demonstrated MP left 25% and right 40% to 50%. Her orthopedist informed Caroline that if the MPs increased, Briella would be a candidate for reconstructive surgery, a pelvic osteotomy (surgical procedure to reshape the acetabulum), and possible varus femoral osteotomy (surgical procedure to realign the proximal femur). But there was also a less extensive surgery, a bilateral hemiepiphysiodeses. Also known as guided growth, this is a procedure where a screw is inserted into the bone enabling it to guide the alignment as the bone grows to allow better coverage over time. There was a limited timeframe in which the hemiepiphysiodeses could be done for it to provide any benefit to Briella due to her growth potential. The orthopedist explained that the osteotomies could still be an option if the hemipiphysiodeses did not provide adequate coverage over time.
Caroline wanted to find out more before considering surgery. She reached out on social media for information about treatment options. A physical therapist responded to ask if they had tried a standing program with Briella. They had asked about getting a stander for Briella when she was 2-3 years old, but her therapist at that time felt getting her to use her walker more would be more beneficial for her than a stander. After the suggestion of a standing program from social media, Caroline researched the benefits of standing for hip health.
Seeking a Standing Program
Standing in a standing frame in hip abduction has been shown to maintain and possibly reduce MP in children with CP. Studies in Sweden have shown that standing in 15 degrees to 30 degrees of hip abduction bilaterally for 10 hours per week reduces MP and preserves range of motion for up to 7 years.9,10 Another study from Spain showed that standing may enhance acetabular development in young children.11
After completing her investigation, Caroline decided to pursue obtaining a stander with lower extremity abduction for Briella. She felt that there was enough evidence that standing can affect hip status, and if a standing program could prevent surgery it was worth a try. Caroline worked with Briella’s therapist and the local DME vendor to obtain a stander through their insurance. Briella received her stander with bilateral hip abduction adjustments in January 2021, and she gradually built up her tolerance to 2 hours per day separated into 2 one-hour sessions, standing in 60 degrees of total abduction. She also continued her daily therapy routines at home and weekly at school.
Briella returned to the orthopedist in April 2021. At that visit, her x-ray showed left 36% and right 32%. Her MPs had remained stable (with some improvement) over the past 6 months. The orthopedist reminded them that Briella would be a candidate for a pelvic osteotomy and possible varus femoral osteotomy if the MPs increased, as the window for the guide growth procedure had closed.
At her October 2021 visit, Briella’s MPs continued to remain stable, and the orthopedist told Caroline that there was no need for surgical intervention at this time. That was music to her ears. By adding standing in abduction to Briella’s treatment routine, her hips had stabilized and she would not need corrective surgery. Her family has also seen an improvement in her posture, upper body strength, and increase in bowel function since the start of her standing program. Briella’s gait and lower extremity range of motion have also shown improvements since using the stander. Use of a standing frame was shown to improve quality of gait in one small study, but the improvements decreased when the standing program was stopped.12
Briella continues her standing program, and the latest x-ray showed the hip MPs continue to be stable with no progression and no need for surgery.
The evidence that standing can affect hip health is limited and the available studies do not fall into the high level of evidence category due to their size and design. The population does not lend itself to large cohorts or randomized controlled trials. The evidence that is available does show that standing can make a difference and, in this case, prevent surgery.
Maryann M. Girardi, PT, DPT, ATP, is a physical therapist and assistive technology practitioner. She has over 40 years of experience working with adults and children with neuro-disabilities in home, school, and acute rehabilitation care settings. She is the Clinical Education Specialist at Altimate Medical Inc, manufacturers of standing and bath safety equipment. For more information, contact [email protected].
Follow Briella and Caroline at @briellaandme on Instagram, Twitter, and TikTok to learn more about Briella’s therapies and standing program.
References
1. Morton RE, Scott B, McClelland V, Henry A. Dislocation of the hips in children with bilateral spastic cerebral palsy, 1985-2000. Dev Med Child Neurol. 2006;48(7):555-558.
2. Soo B, Howard JJ, Boyd RN, et al. Hip displacement in cerebral palsy. J Bone Joint Surg Am. 2006;88(1):121-9.
3. Palisano R, Rosenbaum P, Walter S, Russell D, Wood E, Galuppi B. Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev Med Child Neurol. 1997;39(4):214-223.
4. O’Donnell M, Mayson T, Miller S, et al. Hip Surveillance in Cerebral Palsy. American Academy for Cerebral Palsy and Developmental Medicine; 2017. Available at https://www.aacpdm.org/publications/care-pathways/hipsurveillance. Accessed May 10, 2022.
5. Reimers J. The stability of the hip in children. A radiological study of the results of muscle surgery in cerebral palsy. Acta Orthop Scand Suppl. 1980; 184:1–100.
6. Shore B, Spence D, Graham H. The role for hip surveillance in children with cerebral palsy. Curr Rev Musculoskelet Med. 2012;5(2):126-134.
7. Hägglund G, Alriksson-Schmidt A, Lauge-Pedersen H, Rodby-Bousquet E, Wagner P, Westbom L. Prevention of dislocation of the hip in children with cerebral palsy: 20-year results of a population-based prevention programme. Bone Joint J. 2014;96-B(11):1546-1552.
8. Shore BJ, MulpuriK, Shrader MW, Radiographic Hip Screening in Cerebral Palsy: Developing POSNA-Wide Consensus. J Pediatr Orthop.2021;3(1). Available at: www.jposna.org/ojs/index.php/jposna/article/view/231. Accessed May 10,2022.
9. Martinsson C, Himmelmann K. Effect of weight bearing in abduction and extension on hip stability in children with cerebral palsy. Ped Phys Ther. 2011, 150-157.
10. Martinsson C, Himmelmann K. Abducted Standing in Children With Cerebral Palsy: Effects on Hip Development After 7 Years. Pediatr Phys Ther. 2021;33(2):101-107.
11. Macias-Merlo L, Bagur-Calafat C, Girabent-Farrés M, A Stuberg W. Effects of the standing program with hip abduction on hip acetabular development in children with spastic diplegia cerebral palsy. Disabil Rehabil. 2016;38(11):1075-1081.
12. Salem Y, Lovelace-Chandler V, Zabel RJ, McMillan AG. Effects of prolonged standing on gait in children with spastic cerebral palsy. Phys Occup Ther Pediatr. 2010;30(1):54-65.